In 1879, inventor Thomas Edison perfected the practical electric light bulb at his Menlo Park laboratory in New Jersey. Modern materials scientists now say Revisiting Edison’s Lab reveals that graphene — a one-atom-thick carbon sheet first isolated in 2004 — may have unknowingly formed during those experiments, showing how scientific discovery depends not only on technology but also on theory and interpretation.
Table of Contents
Revisiting Edison’s Lab
| Key Fact | Detail |
|---|---|
| Edison’s experiments | Carbonized plant fibers heated in low oxygen to create filaments |
| Nature of graphene | Single atomic layer of carbon atoms arranged in hexagonal lattice |
| Modern discovery | Isolated in 2004 by Andre Geim and Konstantin Novoselov |
| Why not discovered earlier | Lack of microscopes, quantum theory, and modern materials science |
Today researchers continue studying graphene for next-generation electronics and energy systems. Revisiting Edison’s Lab highlights a recurring lesson in science: discoveries do not occur when materials first appear — they occur when humanity becomes capable of understanding what it is seeing.
How Revisiting Edison’s Lab Relates to the Light Bulb
Edison’s breakthrough depended on creating a filament that could glow without burning. Documentation compiled by the Edison Papers Project shows that researchers tested more than 6,000 plant samples and materials.
His team eventually used carbonized fibers, including bamboo imported from Japan.
Carbonization involves heating organic material in near-vacuum conditions. Hydrogen and oxygen escape, leaving a carbon skeleton. Under high temperatures, the atoms rearrange into layered carbon crystals similar to graphite.
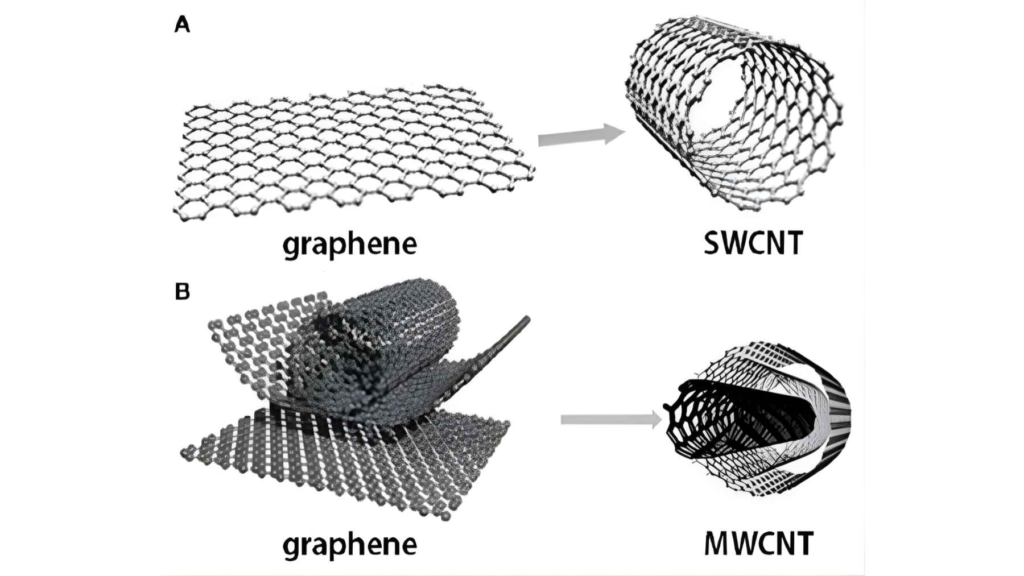
Modern materials science shows graphite consists of stacked atomic layers.
Graphene is simply one isolated layer from that stack.
Scientists studying historical filaments in recent decades have observed graphitic microstructures nearly identical to those seen in modern laboratory carbon materials.
Dr. Mildred Dresselhaus of the Massachusetts Institute of Technology once explained in a lecture on carbon nanomaterials that graphene “is not a new substance, but a new structural form of carbon that has always existed inside graphite.”
This makes Edison’s experiments particularly significant.
He was not merely inventing lighting.
He was unknowingly performing early nanomaterial processing.
The Daily Work Inside Menlo Park
Historical records describe Menlo Park as one of the first industrial research laboratories. It operated around the clock.
Workers:
- shaved filaments
- polished carbon contacts
- cut graphite electrodes
- tested electrical conductivity
The procedures closely resemble modern carbon material preparation techniques.
Today, laboratories intentionally heat organic materials in controlled atmospheres to produce carbon nanostructures. The 19th-century process was primitive but chemically similar.
That is why scientists think graphene flakes almost certainly appeared.
Why Scientists Believe Graphene Likely Formed
During filament production, carbon fibers shrank and cracked. Technicians repeatedly trimmed them to fit lamp holders. These actions mechanically separated carbon layers.
The first confirmed isolation of graphene in 2004 used adhesive tape to peel layers from graphite.
The similarity is striking.
The Nobel Prize Committee noted graphene was obtained through “simple mechanical exfoliation.” Edison’s workers, without realizing it, performed a comparable mechanical process.
Microscopic flakes would have been invisible to the human eye and impossible to identify using Victorian instruments.
The Missing Ingredient: Theory
Atoms Were Still Debated
In the 1870s, atoms were not universally accepted scientific reality. Some physicists still considered them mathematical abstractions.
Without atomic theory, the concept of a one-atom-thick material was almost meaningless.
The scanning tunneling microscope, capable of imaging individual atoms, was invented only in 1981 — more than a century later.
No Solid-State Physics
The discipline of solid-state physics emerged in the early 20th century alongside quantum mechanics.
Edison measured brightness and resistance. Graphene’s importance lies in quantum electron motion — a concept unknown before 1920.
Physicist Antonio Castro Neto later explained graphene behaves as if electrons move at relativistic speeds within the material. No 19th-century instrument could detect such behavior.
Instruments Edison Did Not Have
Three modern technologies were essential to graphene’s recognition:
- Electron microscopy (1930s)
- Quantum theory (1900-1930)
- Atomic-scale imaging (1980s)
Without them, graphene would appear indistinguishable from soot or damaged graphite.
Why Discovery Requires More Than Production
Historians of science emphasize discovery is not simply making something. It is understanding it.
Edison’s laboratory possessed the chemical capability to create graphene-like structures but lacked the interpretive framework.
Many scientific breakthroughs follow this pattern:
- radiation exposed photographic plates before radioactivity theory
- ancient metallurgy created steel before atomic bonding was known
- astronomical observations existed before gravitational theory
The Menlo Park case fits the same historical pattern.
The Broader Context: The Race for Electric Light
Edison was not alone in working on electric lighting. British inventor Joseph Swan was developing carbon filament lamps at nearly the same time.
Both relied on carbonized materials.
This means multiple laboratories across the world may have unknowingly produced graphene-like structures during the 1870s and 1880s.
However, their goal was illumination — not material science.
What Makes Graphene Important Today
Graphene has attracted global attention because of its extraordinary properties:
- stronger than steel by weight
- highly conductive
- flexible
- transparent
- excellent heat conductor
These qualities make it valuable for:
Electronics
Researchers are developing ultrafast transistors and flexible displays.
Energy Storage
Graphene batteries and supercapacitors could charge faster and last longer than lithium-ion batteries.
Medical Sensors
Ultra-thin sensors may detect disease markers in blood.
Aerospace Materials
Lightweight conductive composites could improve aircraft efficiency.
Governments and universities worldwide fund large research programs into nanotechnology and advanced materials.
Economic and Technological Implications
Experts say graphene may influence industries much like silicon did in the 20th century.
Semiconductors built the computer age. Graphene could influence flexible electronics, wearable devices, and next-generation communication systems.
However, large-scale manufacturing remains difficult. Producing uniform, defect-free sheets remains one of the biggest challenges.
What If Edison Had Recognized It?
Historians caution against assuming history would have changed dramatically. Even if Edison noticed unusual carbon films, he lacked theoretical tools to interpret them.
Electronics itself depended on later discoveries:
- electron theory
- vacuum tubes
- transistors
Graphene’s practical applications require microfabrication technology developed only in the late 20th century.
Therefore, recognition in 1879 would not likely have created modern electronics earlier.
Historical Perspective
The comparison between Edison and modern graphene research illustrates how scientific progress works.
Technology alone does not create discovery. Recognition requires understanding.
A laboratory can unknowingly produce a revolutionary material decades before science can identify it.
Scientific knowledge advances when tools, theory, and observation converge at the same time.
FAQs About Revisiting Edison’s Lab
Did Edison discover graphene?
No. There is no evidence he recognized a new material.
Could graphene physically exist in 1879?
Yes. Carbon filament production could generate microscopic graphene flakes.
Why wasn’t it identified?
Scientists lacked microscopes, quantum physics, and nanomaterial theory.
Was Edison close to modern nanotechnology?
Chemically, yes. Scientifically, no.