If you’ve ever watched your phone battery slide from 40% to 5% in what feels like minutes, you’ve experienced the quiet frustration of modern technology. Devices are smarter than ever, electric cars are becoming common, and renewable energy is expanding rapidly yet the battery, the very heart of all of it, still struggles to keep up.

Engineers have squeezed impressive performance from lithium-ion batteries, but they are approaching their natural limits. That is why researchers around the world are paying close attention to one promising development: Breakthrough Sulfur Chemistry May Lead to Longer-Lasting Batteries. This Breakthrough Sulfur Chemistry May Lead to Longer-Lasting Batteries could reshape how we power our daily lives. Instead of relying on expensive metals and complicated materials, scientists are turning to sulfur — a simple, abundant element. Recent laboratory work shows that a new chemical approach can stop sulfur batteries from degrading quickly. The goal is straightforward but powerful: batteries that last years longer, hold more charge, and cost less to produce. For electric vehicles, renewable power storage, and even everyday electronics, the implications are enormous.
The concept behind Breakthrough Sulfur Chemistry May Lead to Longer-Lasting Batteries focuses on lithium-sulfur batteries, often shortened to Li-S batteries. Researchers have studied them for decades because they promise extremely high energy storage capacity. In theory, a lithium-sulfur battery can store two to three times more energy than today’s lithium-ion batteries. However, a major obstacle has always prevented commercialization. During normal charging and discharging, sulfur breaks into intermediate compounds that dissolve inside the battery. This causes rapid capacity loss and short lifespan. The new chemistry stabilizes sulfur at the molecular level so it remains attached to the electrode instead of floating freely. The result is dramatically improved durability and efficiency, opening the door to real-world use in vehicles, electronics, and large-scale energy storage systems.
Table of Contents
Breakthrough Sulfur Chemistry May Lead to Longer-Lasting Batteries
| Feature & Property | Conventional Lithium-Ion Battery | Sulfur-Based Lithium-Sulfur Battery |
|---|---|---|
| Energy Density | Moderate capacity | Very high capacity (up to 3× potential) |
| Battery Lifespan | Degrades gradually | Much slower degradation |
| Material Cost | Uses cobalt & nickel | Uses low-cost sulfur |
| Environmental Impact | Mining intensive | Lower environmental footprint |
| Weight | Heavier packs required | Lighter batteries possible |
| Electric Vehicle Range | Limited by size | Potentially far longer range |
| Safety | Thermal runaway risk | Potentially safer chemistry |
| Grid Storage Suitability | Expensive scaling | More economical for large storage |
Energy storage is the foundation of modern technology. Smartphones, electric transportation, and renewable electricity all depend on efficient batteries. Incremental improvements have helped lithium-ion batteries evolve, but a major leap requires a new chemistry. The Breakthrough Sulfur Chemistry May Lead to Longer-Lasting Batteries may represent that leap. By stabilizing sulfur molecules, scientists have solved the central problem that blocked lithium-sulfur development for decades. If the technology continues progressing, the next generation of devices could run longer, electric cars could travel farther, and renewable energy could become far more reliable. One of the simplest elements on Earth may ultimately power some of the most advanced technologies of the future.
Why Sulfur Matters
- Sulfur may not sound like a high-tech material, but in battery science it is almost ideal. It is abundant, inexpensive, and already produced in massive quantities as an industrial byproduct. Unlike cobalt and nickel, sulfur does not require extensive mining operations or complex supply chains.
- From a scientific perspective, sulfur has an extremely high capacity for storing lithium ions. More lithium ions mean more stored electricity. That alone makes lithium-sulfur technology attractive, but there’s another important benefit: weight. Sulfur is lightweight, allowing batteries to store more energy without becoming heavier.
- For electric vehicles, this could mean dramatically longer driving distances. A car that currently travels 400 kilometers on a charge could potentially exceed 800 kilometers using sulfur-based cells. That is why the Breakthrough Sulfur Chemistry May Lead to Longer-Lasting Batteries is receiving serious attention from automakers and energy companies.
The Polysulfide Shuttle Problem
The challenge that delayed lithium-sulfur batteries for years was a chemical instability known as the polysulfide shuttle effect Inside a lithium-sulfur battery, sulfur undergoes reactions when electricity flows. During this process, sulfur forms intermediate compounds called polysulfides. These compounds dissolve into the battery’s liquid electrolyte and begin moving between electrodes. Instead of staying in place, the material migrates across the cell.
This creates several issues:
- Loss of active battery material
- Reduced charging efficiency
- Chemical side reactions
- Shortened lifespan
Within a relatively small number of charge cycles, early lithium-sulfur batteries lost most of their storage capacity. For real-world applications like electric vehicles, this was unacceptable. A car battery needs to last years, not months. Essentially, sulfur could store plenty of energy, but it could not remain stable long enough to be practical.
A New Molecular Design
- Researchers solved the problem not by building stronger containers, but by changing the chemistry itself. The Breakthrough Sulfur Chemistry May Lead to Longer-Lasting Batteries relies on stabilizing sulfur molecules, so they remain chemically bonded within the electrode.
- Instead of forming loose polysulfides, the sulfur is locked into a structured configuration. It still participates in energy storage reactions, but it no longer dissolves into the electrolyte.
- A simple way to imagine it is to think of sand and bricks. Loose sand shifts and scatters easily. But when you bind it into bricks, it becomes stable and useful. The same concept applies here sulfur is still active, but no longer mobile.
- By preventing the polysulfide shuttle effect, the battery retains its material and maintains consistent performance across many charging cycles.
Performance Results
Early testing has produced encouraging results. The stabilized sulfur batteries show strong capacity retention even after repeated charging and discharging.
Key improvements include:
- Higher energy storage capability
- Longer operational lifespan
- Improved charging efficiency
- Reduced performance decline
Previous lithium-sulfur batteries often failed after fewer than 100 cycles. The new design demonstrates hundreds, and researchers expect future optimization to push that number much higher. Because of its high energy density, the Breakthrough Sulfur Chemistry May Lead to Longer-Lasting Batteries could also significantly reduce battery size. Smaller batteries mean lighter laptops, slimmer phones, and more efficient electric vehicles.
Implications For Electric Vehicles And Renewable Energy
Battery technology is one of the main factors controlling the growth of electric transportation. Consumers often worry about driving range, charging time, and battery replacement cost. Sulfur-based batteries address all three.
- First, higher energy density increases range. Second, lighter batteries improve efficiency. Third, cheaper materials lower production costs.
- The Breakthrough Sulfur Chemistry May Lead to Longer-Lasting Batteries could make electric cars more affordable and practical for long-distance travel.
- Renewable energy systems may benefit even more. Solar panels generate electricity during the day, while demand peaks at night. Wind turbines produce power inconsistently. Large-scale energy storage is essential for reliable clean energy infrastructure.
- Long-lasting sulfur batteries could store excess renewable electricity and release it when needed, helping stabilize power grids.
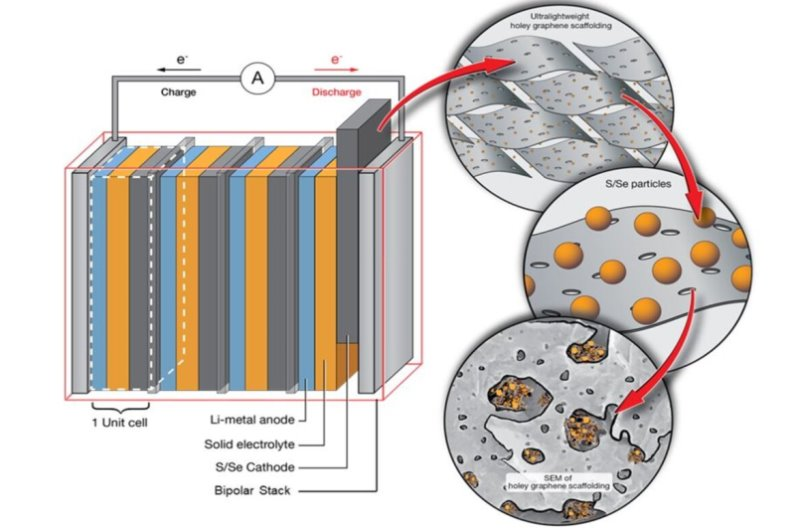
Environmental And Economic Advantages
One major criticism of lithium-ion batteries is their reliance on mined metals. Extracting cobalt and nickel can be expensive and environmentally damaging. Sulfur offers a different path.
Advantages include:
- Reduced mining dependence
- Lower manufacturing costs
- Stable material supply
- Smaller environmental impact
Because sulfur already exists as an industrial byproduct, using it in batteries actually turns waste into a valuable resource. The Breakthrough Sulfur Chemistry May Lead to Longer-Lasting Batteries therefore supports both technological progress and sustainability goals.
Challenges Ahead
Despite strong research progress, commercialization will still take time.
Manufacturers must address:
- Large-scale production methods
- Long-term reliability testing
- Fast-charging capability
- Compatibility with existing battery systems
Batteries must also withstand temperature changes, vibration, and thousands of cycles before reaching consumers. Historically, battery breakthroughs take several years to move from laboratory to market. However, growing investment from automotive and energy industries suggests real confidence in this technology.
FAQs About Breakthrough Sulfur Chemistry May Lead to Longer-Lasting Batteries
1. What makes lithium-sulfur batteries different from lithium-ion batteries?
Lithium-sulfur batteries use sulfur as the cathode material instead of metal oxides. This allows much higher theoretical energy storage capacity.
2. Why were sulfur batteries not used earlier?
They degraded quickly due to the polysulfide shuttle effect, which caused material loss and short lifespan.
3. How does the new chemistry improve battery life?
It chemically stabilizes sulfur molecules so they remain fixed within the electrode and do not dissolve into the electrolyte.
4. Will electric vehicles benefit from this technology?
Yes. Vehicles could achieve longer range, lighter battery weight, and potentially lower cost.